Introduction to Binders for Electrode Manufacturing
Lithium-ion batteries are being promoted as an environmentally-friendly alternative to fossil based fuels for powering vehicle drive trains and also for the generation and storage of electric power. In conjunction with this philosophy, Lithium-ion battery manufacturers are adopting cleaner processes for the fabrication of Lithium-ion electrodes and cells. As a result, water based aqueous binders have gained significant interest in the electrode manufacturing process as an alternative to traditional binder systems. PVDF (Polyvinylidene Fluoride) is one such traditional binder system that has seen the most significant adoption for use as a binder in the lithium-ion battery electrode fabrication process. In such a fabrication process, NMP (N-Methyl-2-pyrrolidone) is generally mixed as a solvent with the PVDF powder. Due to the high cost of NMP, the environment protection issue associated with NMP retrieval, and the stringent processing requirements such as controlling the relative humidity (less than 2%) during the fabrication process, aqueous binders such as SBR (Styrene-Butadiene Rubber) are starting to gradually replace PVDF binders in the fabrication of Li-ion cells in recent years.Hydrophilic Binders vs Conventional SBR Formulations
At present, SBR/CMC composite agent is generally used in the aqueous binder – SBR (Styrene-Butadiene Rubber) as a primary binder and CMC (Sodium Carboxyl Methyl Cellulose) as a thickening/setting agent. The SBR/CMC composite binder has started seeing substantial application in the anodes of lithium-ion cells, however, there are existing bottlenecks in the processing requirements for its application in the preparation of cathode slurries. Aiming at meeting these new processing requirements of environment protection and enhanced cell capacity, a Hydrophilic binder (Modified SB Copolymer) has been developed to overcome the shortcomings of the conventional formulation of SBR/CMC composites and their applicability in the cathode fabrication. In addition to the cost benefits and the pollution-free fabrication process, the Hydrophilic binder offers the following benefits: • Enhancement of the active material ratio in a cell owing to the reduction of the binder amount used • No special requirement for humidity control • Fast drying speeds during the electrode fabricationHydrophilic Binder Characteristics
• Use of a water solvent – environmental friendliness, cleanliness and totally pollution-free • Reduction of usage amount in the cathode slurry – only about 1.5% for cathode and 4% for anode is needed, which enhances the overall cell capacity • No need of environment control in the preparation – no recovery equipment is needed, resulting in lower facility requirements & costs. • Fast drying speed & lower drying temperature decreases electric power consumption and advances electrode production rate by 1.2 ~ 1.5 times • Increasing electrode flexibility, thus facilitating the winding operation in the cell assembly • Following the electrode fabrication, no special dry room treatment in the cell assembly processes • Applicability for both the open and closed formation of lithium-ion cells • No detectable difference in the electrochemical property as compared to the PVDF binderHydrophilic and PVDF Binder Processing Requirements Compared
| Conventional PVDF/NMP Binder | Hydrophilic Binder | ||
| Slurry Formation | LiCoO2 | 93.0% | 94.5% |
| Conducting Carbon | 4.0% | 4.0% | |
| Binder content | 3.0% | 1.5% | |
| Slurry content | 63 – 72% | 65-75% | |
| Humidity control during mixing | Relative humidity control recommended | No humidity control required. Room temperature operation | |
| Coating | Drying Temperature | 90-125C | 45-60C |
| Electrode Drying Time | 6-8 minutes depending on furnace temperature setting | 2-4 minutes | |
| Assembly | Electrode Pressing & Slitting | Relative humidity controlled environment | Room temperature environment – no humidity control required |
| Winging & Enclosure | Relative humidity controlled environment | Room temperature environment – no humidity control required | |
| Electrolyte Injection | Dry environment | Dry environment | |
| Formatting Conditions | Applicable for closed formation | Applicable for closed and/or open formations | |
Hydrophilic and PVDF Binder Performance Comparison
The figures below show the electrochemical characteristics of coin cells created using Hydrophilic and PVDF binders, respectively: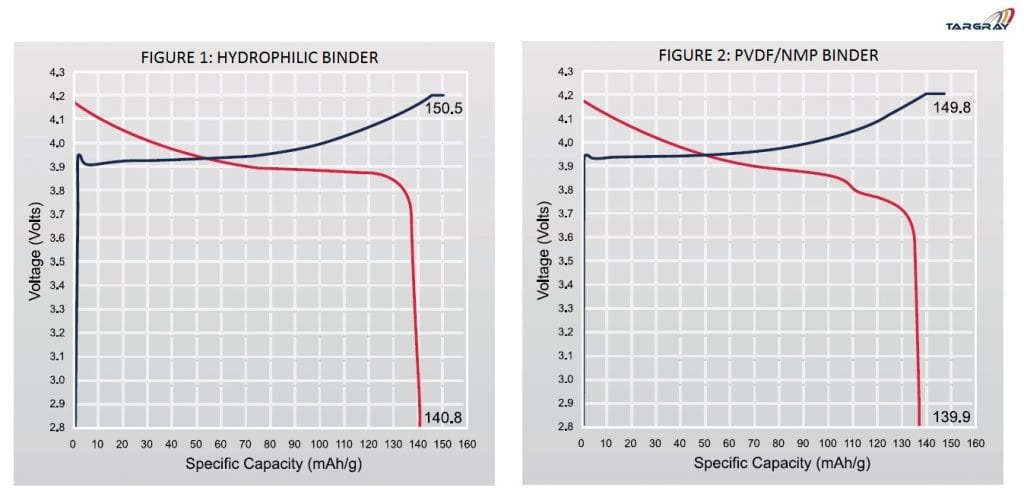 Fig. 1 above depicts the charge and discharge curves of a cathode made using the Hydrophilic binder and cathode material and Fig. 2 depicts the charge and discharge curves of another cathode comprising of similar cathode material but using the PVDF binder. As compared to the performance of the cathode using PVDF binder, the adoption of Hydrophilic binder is capable of delivering completely the capacity of the cathode material (approximately 140mAh/g as illustrated above).
Fig. 1 above depicts the charge and discharge curves of a cathode made using the Hydrophilic binder and cathode material and Fig. 2 depicts the charge and discharge curves of another cathode comprising of similar cathode material but using the PVDF binder. As compared to the performance of the cathode using PVDF binder, the adoption of Hydrophilic binder is capable of delivering completely the capacity of the cathode material (approximately 140mAh/g as illustrated above).Conclusion
Using Hydrophilic binders (Modified Styrene-Butadiene Copolymer) in Lithium-ion electrode fabrication can lead to simpler, cleaner and more cost-effective cell processing. It also enables battery technology developers to achieve higher cell capacity while using less binder material. These unique advantages help reverse the traditionally held belief that hydrophilic binders are ill-suited for cathode fabrication.Targray is one of the fastest-growing suppliers of lithium-ion battery materials and chemicals in the world. Visit the Targray Battery page for more information on the products we offer, including both Hydrophilic & PVDF binders.